You’d think there’s nothing surprising left to discover about water. After all, researchers have been studying its properties for centuries.
But today researchers at the Department of Energy’s Pacific Northwest National Laboratory report a new finding. Even though ice forms in a perfectly hexagonal lattice, it is surprisingly flexible and malleable, which explains why ice so often has trapped gas bubbles.
The findings come from the first-ever molecular-resolution observations of nanoscale samples of ice frozen from liquid water, which were published today in the journal Nature Communications.
“We observed dissolved gas not only generate cavities in ice crystals, but also migrate, merge with other gas bubbles and dissolve—behavior that is only possible due to the unusual nature of bonding in ice,” said James De Yoreo, principal investigator of the work and a Battelle Fellow at PNNL. “This work opens up an entirely new opportunity to explore ice crystallization and melting behavior at scales unimaginable only a few years ago.”
The research could have profound implications for preserving deeply frozen (cryogenic) biological tissue samples, forecasting ice behavior for aviation and vehicle safety and understanding the movement of glaciers, among other areas of research.
“There have been a lot of mysteries about ice,” said PNNL materials scientist Jingshan Du, lead author of the work. “We want to understand how ice tolerates structural imperfections in the crystal and how trapped bubbles affect the mechanical properties of the crystal. Now we have a way to understand that.”
What’s new with ice
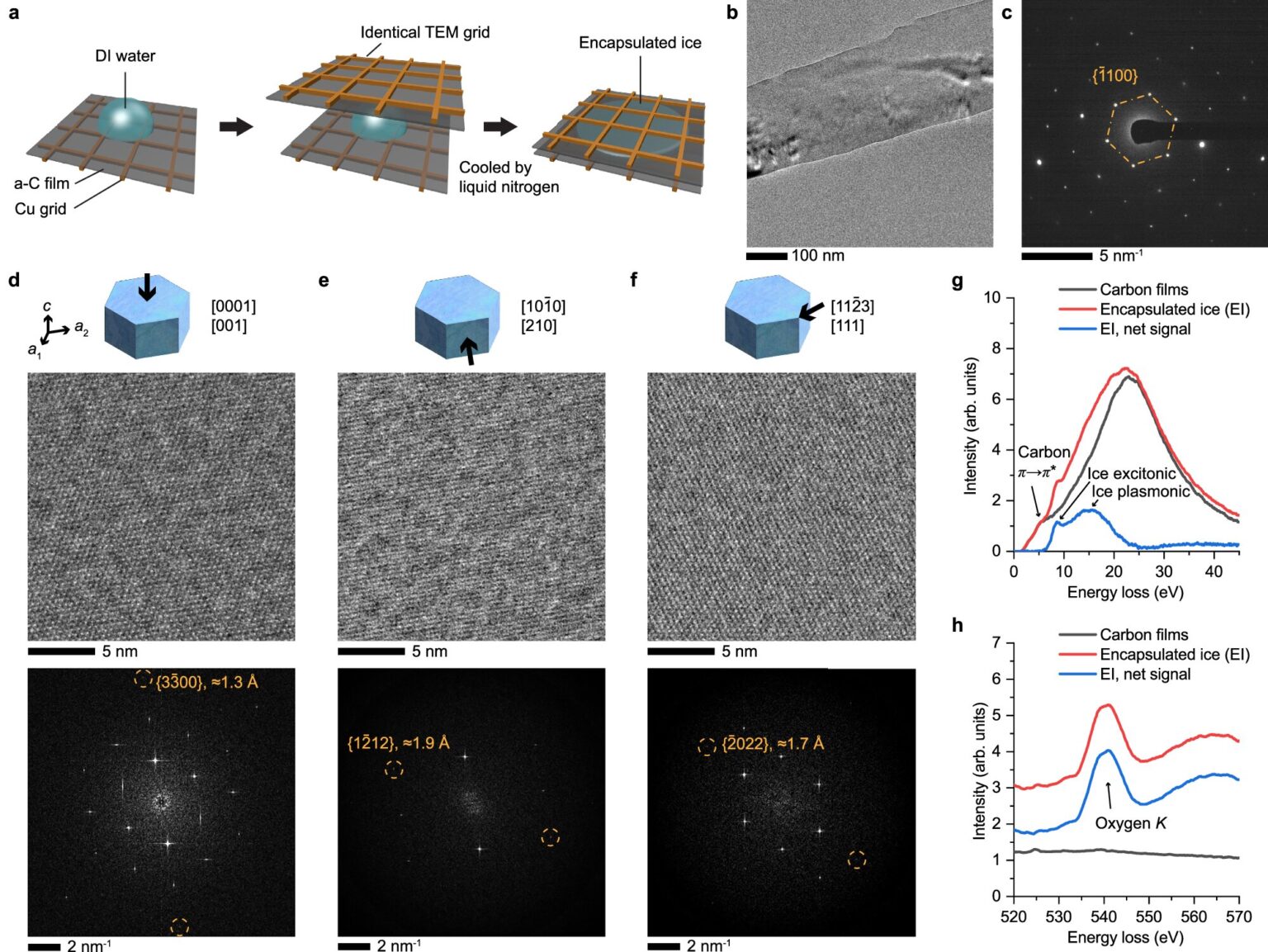
Until now, no one has been able to directly observe molecules of water undergoing the shift from liquid water to ice. That’s because the techniques scientists use to view individual atoms involve harsh conditions, including using high-energy radiation and removal of all air (vacuum sealing). While researchers have generated some images of ice at the molecular scale, that ice doesn’t reflect the normal freeze-thaw cycles on Earth. It’s generated by flash freezing directly from vapor to solid.
To avoid those issues, the research team sandwiched liquid water between thin carbon membranes, which turned out to be the critical factor that led to this imaging breakthrough. Then they developed a new technique, cryogenic liquid-cell transmission electron microscopy, to follow the freezing process.
“The membranes protect the ice crystals from high vacuum and radiation, allowing us to acquire images with atomic-level information,” said Du.
They watched gas bubbles form, move through the lattice, merge with other bubbles and dissolve.
The study results showed that when liquid water turns to solid ice, defects in its crystal structure or trapped gas bubbles don’t cause much strain to the ice crystal, which could cause fracturing. It adapts to the presence of the defect with surprising ease compared with other solids, like metal or minerals.
The nature of water’s chemical bonds makes it extraordinarily flexible and malleable, even as solid ice. This new observation, combined with the crucial fact that ice is less dense than liquid water, are properties that support life on Earth, and especially in the sea.
The researchers also made direct observations of the geometries and forces that guide ice crystal formation at all scales, including the formation of snowflakes. While snow forms from water vapor, not liquid water, the same underlying forces are at work.
To confirm their experimental observations, the PNNL scientists collaborated with researchers from Argonne National Laboratory and the University of Illinois-Chicago who had used machine learning to develop a highly accurate molecular dynamics model for ice. The comparisons between the experiment and what was predicted by theoretical models confirmed that ice is unique among solids in its tolerance for defects without compromising the integrity of the ice crystal structure.
Why trapped air bubbles in ice matter
While the PNNL team is studying ice dynamics on a nanoscale, other researchers are discovering that the presence of air bubbles in glaciers greatly affects their behavior. Recently, scientists showed that glaciers melt more than two times faster if they contain bubbles, compared with bubble-free ice. Other scientists are trying to avoid having ice form in delicate tissue samples or on aircraft in flight.
Next steps for this research include studying melting and working with more complicated samples, including water with dissolved materials.
More information:
Jingshan S. Du et al, Molecular-resolution imaging of ice crystallized from liquid water by cryogenic liquid-cell TEM, Nature Communications (2025). DOI: 10.1038/s41467-025-62451-0. www.nature.com/articles/s41467-025-62451-0
Citation:
A chilling discovery: The surprising flexibility of ice at the nanoscale (2025, September 25)
retrieved 26 September 2025
from https://phys.org/news/2025-09-chilling-discovery-flexibility-ice-nanoscale.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.