Researchers in Japan have created a novel nanocarrier expertise that delivers therapeutic antibodies straight to focus on antigens inside most cancers cells, marking an essential breakthrough towards higher focused most cancers remedy. The findings have been revealed within the Journal of Managed Launch on June 4th, 2025.
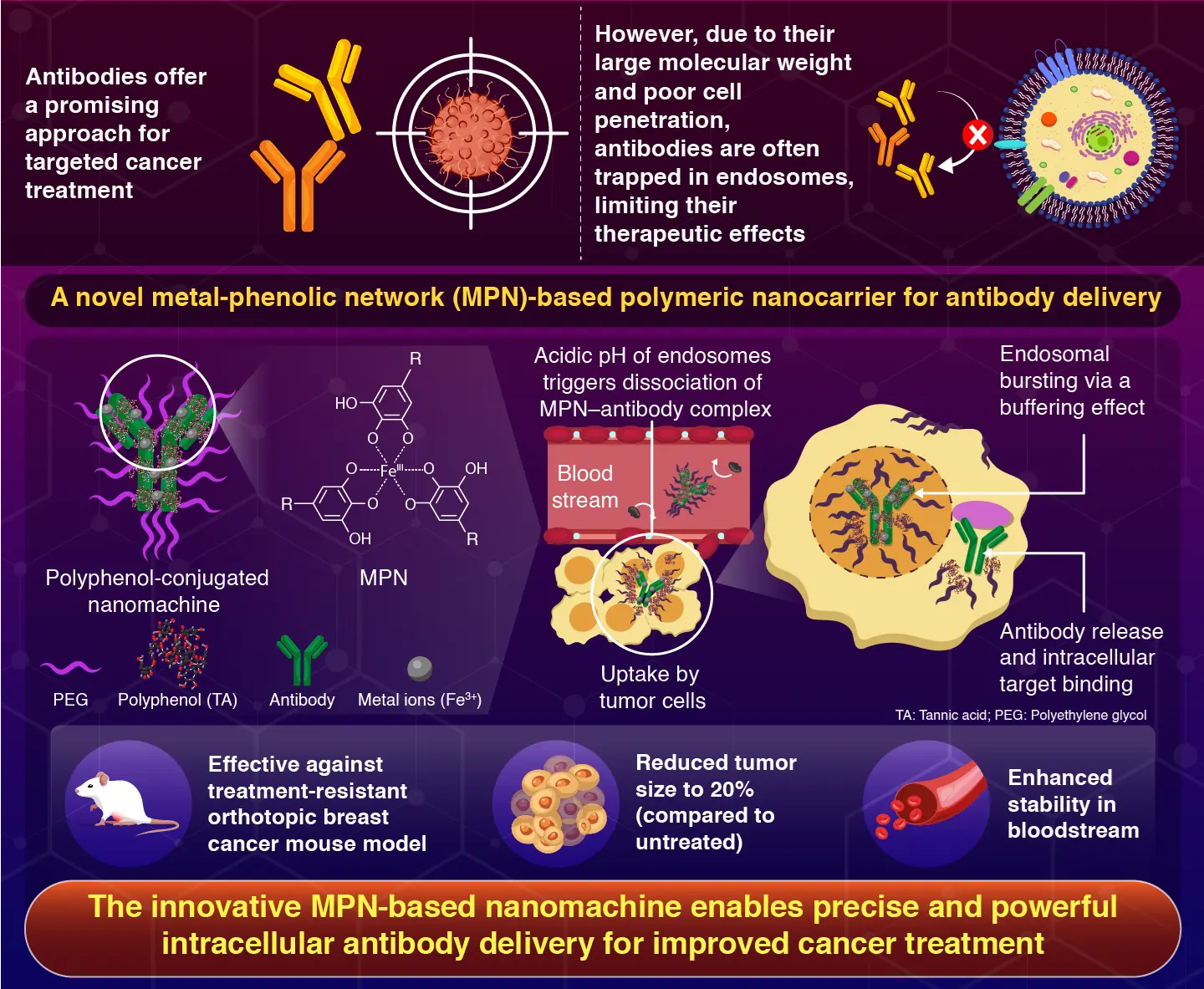
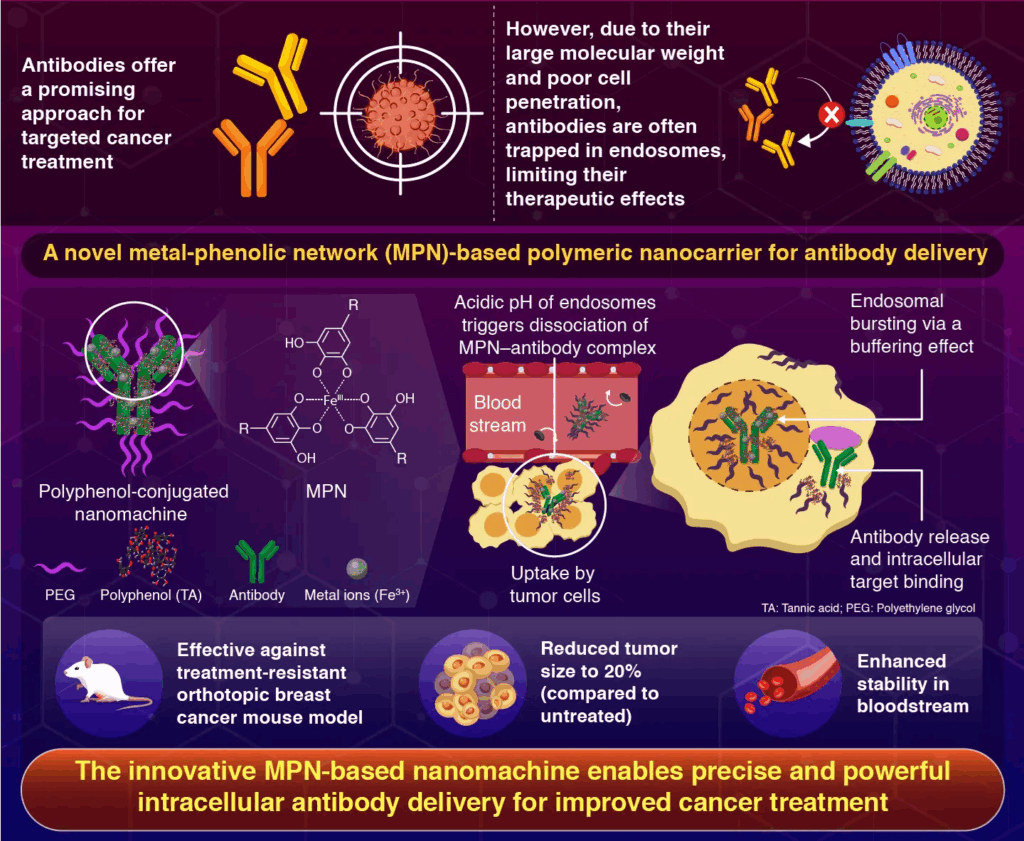
Utilizing a metal-polyphenol community, the system escapes endosomes, permitting for intracellular concentrating on of antibodies, leading to tumor progress inhibition and elevated anti-cancer exercise, making it a viable device for focused most cancers remedy.
Overcoming Mobile Obstacles: Polyphenol-Primarily based Nanocarriers for Antibody Supply
The physique’s immune system produces Y-shaped proteins referred to as antibodies to acknowledge and neutralize invading substances. By figuring out distinct markers or antigens on the tumor surfaces, therapeutic antibodies—specifically designed proteins created from pure antibodies—goal most cancers cells. By defending wholesome tissues, these proteins assist the immune system fight cancers extra efficiently.
Though therapeutic antibodies have been proven to be efficient in opposition to most cancers by concentrating on cell surfaces, their efficacy inside cells is restricted resulting from their incapability to cross cell membranes and escape endosomal entrapment—a course of by which molecules grow to be trapped inside endosomes (membrane-bound compartments) inside cells. To beat this hurdle, researchers at the moment are investigating a number of strategies for stopping antibody endosomal entrapment.
A analysis workforce from the Laboratory for Chemistry and Life Science, Institute of Science Tokyo (Science Tokyo), Japan, led by Assistant Professor Yuto Honda and Professor Nobuhiro Nishiyama, has tackled this downside by utilizing polyphenols, a category of compounds current in wine, to create a novel nanomachine loaded with antibodies.
The Innovation Heart of Nano Medication, a division of the Kawasaki Institute of Industrial Promotion, collaborated within the growth of the expertise.
The examine particulars the creation of a metal-phenolic community (MPN) polymeric nanocarrier that permits therapeutic antibodies to be exactly delivered intracellularly into most cancers cells. The technique makes use of polyphenols, which have a particular mechanism for endosomal escape and antibody distribution.
We developed a nanomachine utilizing polyphenols, polyethylene glycol (PEG), and metallic ions to encapsulate the antibodies. As soon as contained in the cells, the metallic ion-polyphenolic group community triggers a buffer impact which ends up in the bursting of endosomes, releasing the antibodies on the goal web site.
Yuto Honda, Assistant Professor, Institute of Science Tokyo
The researchers started by conjugating tannic acid (TA), a polyphenol compound, with PEG to create PEG-TA, a nanocarrier system. PEG is a polymer that stabilizes the system due to its robust biocompatibility and stealth qualities.
To create an antibody-loaded MPN complicated, the PEG-TA molecules have been mixed with the therapeutic antibody and ferric chloride (Fe3+ metallic ions). Transmission electron microscopy and fluorescence correlation spectroscopy have been used to look at the properties of those nanomachines, which had a diameter of 30 nm.
The administration and mobile absorption effectiveness have been evaluated in vitro, and anti-tumor exercise was confirmed in vivo in an orthotopic mouse mannequin of treatment-resistant breast most cancers. The outcomes demonstrated that the nanomachines have been very secure within the circulation, with elevated absorption by tumor cells, reducing tumor progress by 20% in comparison with the untreated management group. The numerous anti-tumor efficacy was linked to the novel mode of antibody launch.
When the nanomachines are absorbed by the tumor cells, they grow to be entrapped inside endosomes. The MPNs separate from antibodies as a result of endosomes’ inside acidic pH, and the MPNs which are launched produce a buffering impact that encourages the entry of protons and counterions from the skin of the endosome.
This damages the endosome membrane and raises the inner osmotic stress. The dissociated antibodies are launched when the endosomes burst and might subsequently bind to the antigen targets contained in the cell.
The examine revealed the efficient supply of an anti-S100A4 antibody, which restored the perform of the tumor suppressor protein p53, leading to tumor cell dying. Moreover, the consequences have been recognized with low toxicity.
Honda concluded, “Our examine marks a big step towards growing next-generation intracellular antibody therapies. The non-cationic, biocompatible, and systemically injectable design of our MPN system may broaden its functions past most cancers, paving the best way for next-generation drug concentrating on.”
Journal Reference:
Honda, Y., et al. (2025) Steel-phenolic network-based polymeric nanocarriers facilitating antibody cytoplasmic supply and anti-tumor results to orthotopic breast tumors. Journal of Managed Launch. doi.org/10.1016/j.jconrel.2025.113929.
Supply: