A group led by Prof. Horacio Cabral has discovered a new method to construct protein complex-based therapeutics.
Prof. Cabral is a visiting scientist from the Innovation Center for NanoMedicine (iCONM), and is Associate Professor of the Department of Bioengineering, Graduate School of Engineering, University of Tokyo.
The paper, titled “Nano-enabled IL-15 superagonist via conditionally stabilized protein-protein interactions eradicates solid tumors by precise immunomodulation,” was published in the Journal of the American Chemical Society on October 2, 2024.
Protein complexes are critical regulators of diverse biological processes, exhibiting unique functions that make them promising candidates for therapeutic applications. For example, interlukin-15 (IL-15) can bind with its receptor α domain (IL-15Rα) to form a superagonistic complex, which can effectively activate antitumoral immunity by facilitating the trans-presentation of IL-15 to immune cells.
However, protein complexes are mainly formed by dynamic non-covalent interactions, leading to instability and transient effective time in biological environments. Thus, delivery strategies are needed to mediate stable delivery of protein complexes.
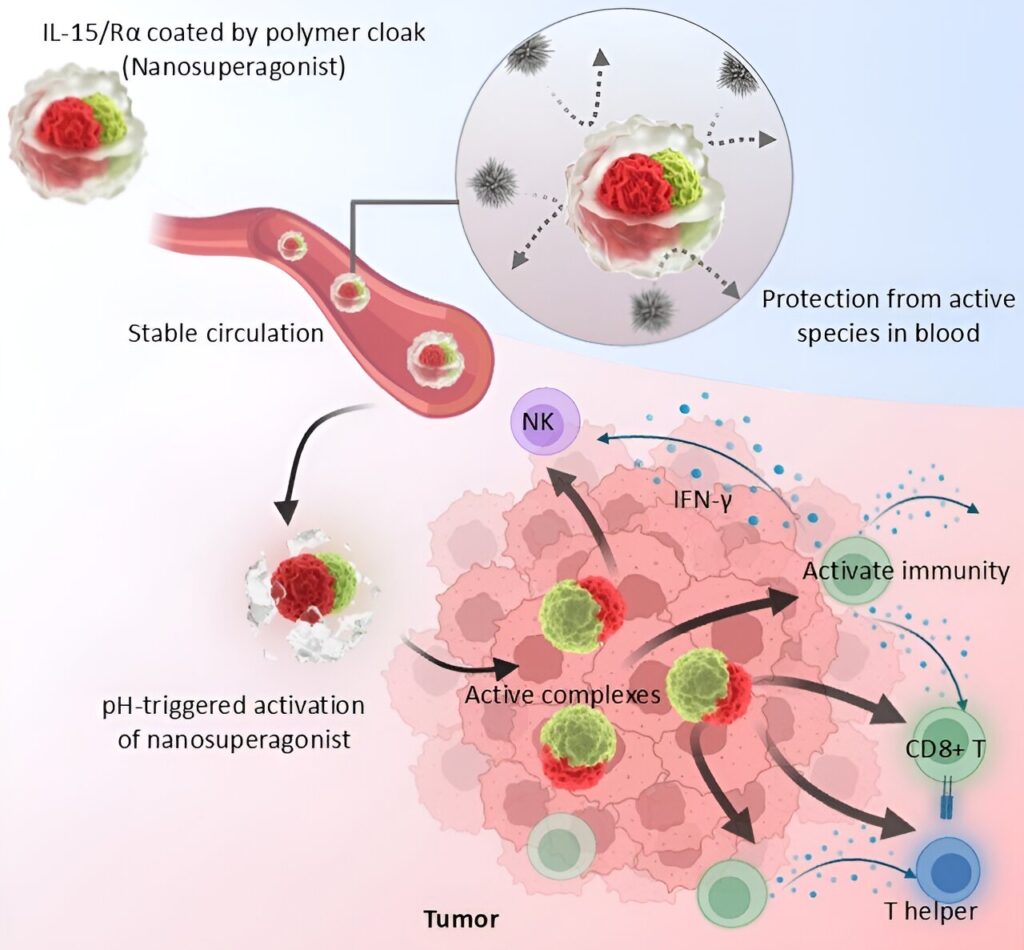
The current study presents a polymer cloak that was designed to assist the use of protein complexes as therapeutic agents, as illustrated by the prototype formulation loading the IL-15/IL-15Rα complex.
The polymer coating stabilized the protein–protein interaction between IL-15 and IL-15Rα, which effectively immobilized the complex, isolating it from the surroundings and yielding an IL-15 nanosuperagonist with enhanced efficacy. In mouse models, the nanosuperagonist displayed improved pharmacokinetics and stably delivered the intact protein complex upon intravenous injection.
Moreover, due to the pH-sensitivity of the polymer cloak, the nanosuperagonist could sense the acidic tumor microenvironment and ensure a tumor-selective activation. In murine colon cancer models, the nanosuperagonist profoundly inflamed the tumor to eliminate the cancer cells without inducing immune-related side effects.
As illustrated in this study, the IL-15/IL-15Rα complex could be disrupted by external reactive species like monomeric IL-15 or proteases, leading to the rapid disintegration of the complex in vivo.
In contrast, the polymer cloak interlocked the complex and protected its integrity in harsh environments like the blood stream. Thus, the polymeric cloak could promote the stability and function of the protein complex in in vivo conditions, which is critical for attaining therapeutic activity.
Compared to the reported methods for delivering protein complexes, which focus on engineering protein structures to create stable fusion proteins, this polymer cloak detours the complexity in design and manufacture of protein engineering technology.
In addition, the system can be easily extended to apply to other protein complex payloads beyond the illustrated IL-15/IL-15Rα complex, serving as a universal platform. Moreover, benefiting from the pH-sensitive nature of the polymer cloak, the system not only mediated a stable delivery of the intact protein complexes systemically, but further achieved a tumor-targeted release of the cargos.
In the case of the IL-15-based nanosuperagonist, the polymer coating stabilized the IL-15/IL-15Rα complex to provide antitumor superagonistic potency, and the pH-controlled de-coating of the polymer allowed a tumor-specific bioactivity, which mitigated the toxicity.
Thus, compared to the conventional fusion protein-based IL-15 superagonist formulations, the nanosuperagonist realized a safer therapy. In summary, the polymer cloak presents a streamlined platform for tumor-targeted delivery of protein complexes, and it will inspire further research into polymer- and nano-based approaches to facilitate the translation of protein complexes as therapeutic agents.
Provided by
Innovation Center of NanoMedicine