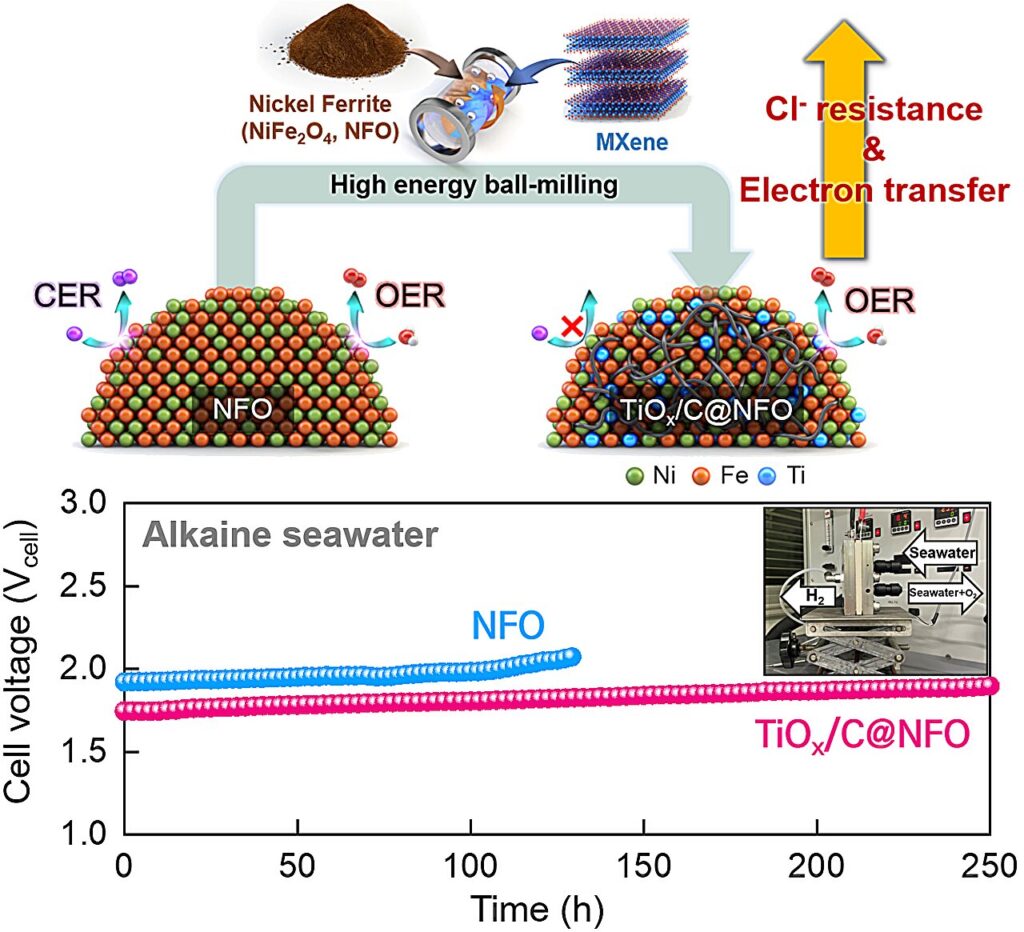
A research team has developed a composite catalyst using the novel material MXene that suppresses the generation of chloride ions—one of the key challenges in seawater electrolysis. This research outcome is expected to accelerate the practical application of seawater electrolysis technology by enabling stable hydrogen production even in seawater.
The findings are published in the journal ACS Nano. The study was led by Dr. Juchan Yang at the Hydrogen & Battery Materials Center, from the Energy & Environment Materials Research Division of the Korea Institute of Materials Science (KIMS).
Hydrogen is gaining attention as an eco-friendly energy source that emits no carbon. However, conventional water electrolysis technologies primarily use clean freshwater, which leads to high production costs and raises concerns over water resource availability. Seawater electrolysis, an alternative that directly uses seawater, has emerged to address these drawbacks. Nonetheless, a critical challenge remains: chloride (Cl⁻) ions present in seawater can easily corrode the electrolysis electrodes, significantly shortening the lifespan of hydrogen production systems.
MXene is a two-dimensional nanomaterial composed of metals and either carbon or nitrogen. It possesses excellent electrical conductivity and can be combined with various metal compounds, making it well-suited for use as an electrode material. However, it has a notable limitation: its high reactivity with oxygen and water makes it prone to oxidation, which hinders its long-term stability and application.
To address this issue, the research team intentionally oxidized the MXene to form a stable conductive structure and fabricated an electrode composite catalyst by combining it with nickel ferrite (NiFe₂O₄), an oxygen evolution catalyst, using a high-energy ball milling process. The resulting composite catalyst exhibited approximately five times higher current density and twice the durability compared to conventional catalysts. In addition, it demonstrated excellent repulsion toward chloride ions, effectively preventing electrode corrosion.
Through this process, the team achieved high uniformity and reproducibility, laying the groundwork for large-scale production. Furthermore, beyond laboratory-scale catalyst performance evaluations, the team successfully validated the material’s performance in an actual electrolysis unit cell, confirming its practical applicability.

This technology is highly significant in that it overcomes the limitations of conventional MXene-based materials by simultaneously securing both conductivity and durability, making it suitable for application in seawater electrolysis electrodes. Moreover, by developing a high-performance electrode material that suppresses corrosion issues in seawater electrolysis, it is expected to accelerate practical implementation and contribute to the global expansion of hydrogen production infrastructure.
Dr. Juchan Yang, the principal investigator at KIMS, stated, “This study is significant in that it addresses the issue of chloride ions in seawater by utilizing the novel material MXene.
“We are actively conducting follow-up demonstration research to further advance this technology into a sustainable hydrogen production solution.”
More information:
Kyung-Bok Lee et al, Durable Seawater Electrolysis through the Synergistic Effect of Oxidized MXene/Nickel Ferrite Composite Electrocatalyst, ACS Nano (2025). DOI: 10.1021/acsnano.5c04312
Citation:
MXene electrode material withstands seawater corrosion in hydrogen production (2025, September 16)
retrieved 20 September 2025
from https://phys.org/news/2025-09-mxene-electrode-material-seawater-corrosion.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.