A team of scientists has found a way to slow energy leaks that have impeded the use of tiny nanocrystals in light-driven chemical and energy applications.
As described in an article published in the journal Chem, the team has used a molecule that strongly binds to the nanocrystal’s surface, essentially acting like a dam to hold back the energy stored in the charge-separated state formed after light absorption. This technique extends the lifetime of the charge separation to the longest recorded for these materials, providing a pathway to improved efficiencies and more opportunities to put this energy to work in chemical reactions.
The researchers from the University of Colorado Boulder, the University of California Irvine, and Fort Lewis College were led by RASEI Fellow Gordana Dukovic.
Harnessing light to power chemistry
Many of the products we rely on today, from plastics, to fertilizers, and pharmaceuticals, are created, or synthesized, through industrial chemical reactions that can often require immense heat and pressure, typically generated by burning fossil fuels. For decades there has been research exploring a less harsh and theoretically more efficient alternative: photocatalysis. The goal is to use a compound, a “photocatalyst,” that can harness the energy in light and use it to power chemical reactions at room temperature.
Semiconductor nanocrystals, particles that are over a thousand times smaller than the width of a human hair, are a leading candidate for this job. When exposed to light, these nanocrystals generate a short-lived spark of energy, in the form of a separate negative charge (an electron) and a positive charge (called a “hole,” due to the absence of an electron). A key challenge in this area is that this spark disappears quickly, because the electron and the hole recombine, and the energy is lost before it can be put to good use.
Building a molecular dam
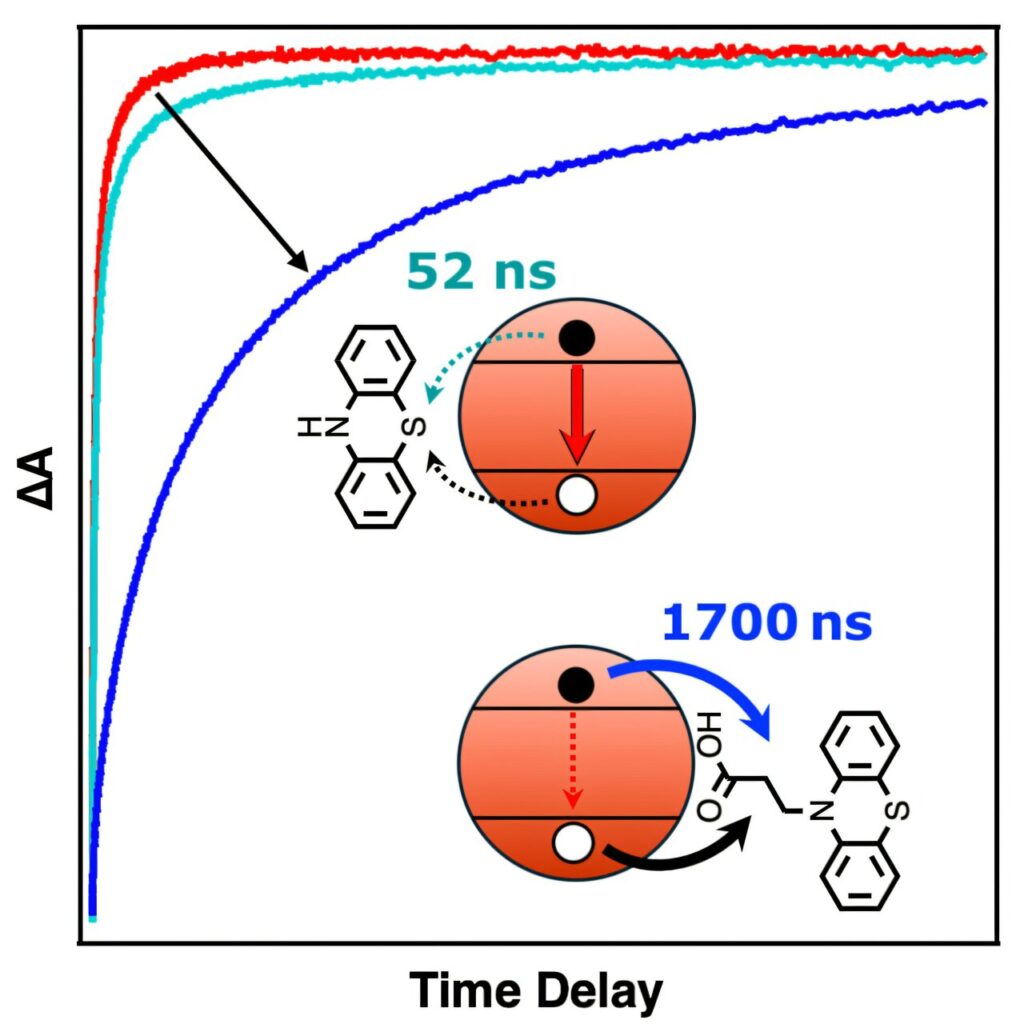
To solve this problem, the team focused on building what we might call a molecular dam, something that helps prevent—or at least slow down—the electron and the hole from recombining. This research started with cadmium sulfide (CdS) nanocrystals and designed a molecule (in this case a phenothiazine derivative) with two key features; first, the incorporation of a chemical group that acts as a sticky anchor (in this case a carboxylate group), which binds strongly to the nanocrystal surface; and second, a molecular structure that quickly accepts the positive charge (the hole), from the nanocrystal to realize the light-driven charge separation event.
By anchoring this molecule to the surface of the nanocrystal, the team created a highly efficient and stable pathway. As soon as exposure to light creates the electron-hole pair in the nanocrystal, the anchored molecule shuttles the hole away, physically separating it from the electron. This physical separation of the electron and the hole prevents the two from quickly snapping back together and wasting energy.
This results in a charge-separated state that lasts for microseconds, which is an eternity in the world of photochemistry, creating a much larger window of time for future researchers to work with in terms of harnessing this captured light-driven energy for useful chemical reactions.
The team was able to prove the significance of the sticky anchor carboxylate, by comparing their derivative to a phenothiazine that lacked the anchor, which was shown to be far less effective at holding the energy, demonstrating that this anchoring to the surface was key to this system’s performance.
This project took advantage of the different areas of expertise of each team to generate ideas and quickly execute them. Kenny Miller’s group of dedicated undergraduate researchers at Fort Lewis College synthesized the carboxylated phenothiazine derivative (and a slew of others).
Miller then sent the derivative to Jenny Yang’s group of inorganic electrochemists at UC Irvine for advanced electrochemical characterization. Gordana Dukovic’s group at CU Boulder synthesized the nanocrystals, tested their compatibility with the derivative, characterized the binding, and undertook the advanced laser spectroscopy study to see how the electrons and holes behaved.
“The first time I saw the results—saw how effective our ‘molecular dam’ was at slowing charge recombination—I knew we had struck gold,” explained Dr. Sophia Click, a lead author on the study. “To slow charge recombination from nanoseconds to microseconds, and with a molecule that can be paired with so many existing photocatalyst systems, makes this work vital to share with as many researchers as possible.”
Development of this molecular dam could have implications for the future design of catalysts for light-driven chemistry. By increasing the efficiency of the initial energy-capture step, this system improves the efficiency of the entire process. This could improve not just one specific reaction, but rather, benefit a broad range of light-driven chemical reactions. A key technology this could enhance is the development of light-driven creation of chemical commodities or high-value chemicals.
This research provides a more robust and versatile chemical toolkit for exploring these possibilities.
This discovery in controlling charge-separation and energy at the nanoscale is an important design parameter in developing light-driven chemistry, and hopefully light-driven chemical manufacturing. Imagine a future where materials, such as plastics, and even pharmaceuticals, are not made in energy inefficient high-temperature reactors powered by fossil fuels, but instead are synthesized directly and efficiently using the power of light.
While this vision is still on the horizon, the work done in this collaboration provides an important piece of the scientific puzzle, constituting a huge leap toward one day achieving these goals.
More information:
Sophia M. Click et al, Exceptionally long-lived charge-separated states in CdS nanocrystals with a covalently bound phenothiazine derivative, Chem (2025). DOI: 10.1016/j.chempr.2025.102760
Citation:
‘Molecular dam’ stops energy leaks in nanocrystals to boost efficiency of light-driven reactions (2025, October 23)
retrieved 24 October 2025
from https://phys.org/news/2025-10-molecular-energy-leaks-nanocrystals-boost.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.